
On December 7, 2025, the 2025 National Reimbursement Drug List and the first edition of the Commercial Insurance Innovative Drug List were released in Guangzhou. This year’s basic medical insurance list shows the greatest support for innovative drugs in recent years. When adjusting the basic reimbursement list, the National Healthcare Security Administration (NHSA) is more willing to support true innovation and differentiated innovation, rather than me too innovation.
Two innovative targeted drugs for breast cancer – Inavolisib and Capivasertib – have been successfully included in the reimbursement scope. The two drugs were approved on March 14 and April 18 of this year, respectively. Their rapid inclusion greatly improves drug accessibility, reflecting the NHSA’s genuine support for true innovation.

This marks that the treatment of HR+/HER2 advanced breast cancer in China has officially entered the era of "precise pathway inhibition" , and also means that genetic testing has become an indispensable "navigation system" for clinical medication. In this context, comprehensive genotyping based on next generation sequencing (NGS) is gradually becoming the cornerstone of personalized treatment for breast cancer.
Breast Cancer NGS Genetic Testing: From "Optional" to "Essential"
Breast cancer is highly molecularly heterogeneous. Different patients have vastly different driver gene mutation profiles, which directly affect treatment response and prognosis. In the past, treatment selection relied mostly on immunohistochemical subtyping. However, with the continuous emergence of drugs targeting the PI3K/AKT/mTOR pathway, PARP inhibitors, CDK4/6 inhibitors, and novel targets, treatment decisions are increasingly dependent on precise genetic information.
In particular, Inavolisib (targeting PIK3CA mutations) and Capivasertib (targeting PIK3CA/AKT1/PTEN mutations), which have now entered the reimbursement list, both explicitly require specific genetic alteration statuses before use. This undoubtedly pushes genetic testing from an auxiliary reference to a clinical necessity.
Why NGS? – Comprehensive, Accurate, Efficient
Among various testing technologies, NGS, with its unique advantages, has become the preferred method for simultaneous multi gene analysis in breast cancer.
1. Comprehensive Coverage, Avoiding Misses
Key breast cancer genes such as PIK3CA and PTEN have complex variation patterns. The Chinese Expert Consensus on PIK3CA Gene Mutation Testing in Breast Cancer (2025 Edition) points out that breast cancer patients with multiple PIK3CA point mutations have higher sensitivity and overall response rates to PI3K inhibitors compared to those with a single point mutation. Therefore, comprehensive detection of PIK3CA mutation sites is strongly recommended.
PIK3CA mutations include single site and multi site mutations. Among PIK3CA mutated patients, about 88% have single site mutations, while 12 15% have multi site mutations. The most common PIK3CA double mutations in breast cancer consist of one hotspot mutation (including E542, E545, or H1047) and a second rare site mutation (including E453, E726, or M1043).
Traditional PCR methods cannot comprehensively cover the 62 PIK3CA mutations or the rare site PIK3CA mutations involved in the Inavolisib clinical trials. PTEN inactivation often involves non hotspot mutations or large fragment deletions, which are difficult to detect by traditional PCR. NGS can detect dozens to hundreds of genes in a single run, covering point mutations, insertions/deletions, copy number variations, gene fusions, and other variant types, greatly reducing the risk of missed detection.

2. One Test, Multiple Benefits – Guiding Multiple Strategies
A single NGS test can not only screen for beneficiaries of the above targeted therapies but also simultaneously assess homologous recombination repair related genes such as BRCA1/2 (guiding PARP inhibitor use), ESR1 mutations (indicating endocrine resistance and SERD class selection), HER2 amplification/mutation, TMB, MSI, and other immunotherapy related biomarkers, as well as hereditary cancer risk – achieving "one sample, multi dimensional evaluation".
3. Aligned with Guidelines, Standardized Approach
Domestic authoritative consensus documents, such as the Clinical Application Guidelines for Genetic Testing in Advanced Breast Cancer (2025 Edition), explicitly state that NGS is the preferred method when conditions permit. Its comprehensiveness better matches the complex biological characteristics of advanced breast cancer, including multi gene co variation and dynamic evolution, providing a solid basis for later line treatment strategies.
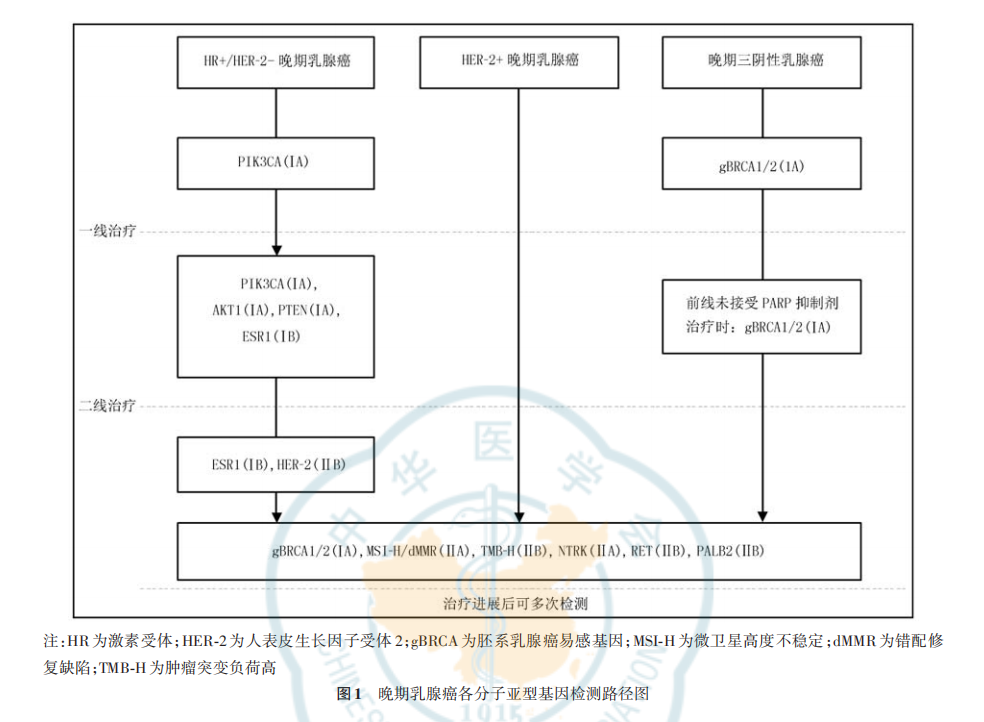
YunKang Breast Cancer NGS Testing: Enabling Precision with Professionalism
YunKang Group · Guangzhou DaAn Clinical Laboratory, relying on a mature NGS platform and rigorous bioinformatics analysis processes, offers a series of products including Breast Cancer 18 Gene Testing, Breast Cancer 65 Gene Testing, and Solid Tumor 1226 Gene Testing (all of which cover PIK3CA/AKT1/PTEN and can guide Inavolisib and Capivasertib targeted therapy), committed to providing reliable and comprehensive genetic testing solutions for clinicians and patients.
Rigorous Quality Control, Reliable Results
From sample pathological assessment, DNA extraction, library construction, to high depth sequencing, a strict quality control system is implemented throughout the entire process to ensure accurate and reproducible data.
Professional Interpretation, Clinically Relevant
An in house interpretation database is dynamically updated weekly, staying close to the forefront of precision medicine. The test report not only lists variant information but also classifies the clinical significance of variants according to domestic and international guidelines and the latest evidence based data, clearly indicating relevant medication information for targeted therapy, chemotherapy, endocrine therapy, and immunotherapy, assisting clinicians in formulating personalized regimens.
Comprehensive Coverage, Focus on Core Genes
The testing covers core breast cancer driver and resistance genes such as PIK3CA, AKT1, PTEN, ESR1, BRCA1/2, HER2, while also assessing HRR status, PD L1 expression, and genetic risk, fully meeting the needs of precision diagnosis and treatment. (Note: Testing scopes vary among different products; please refer to the specific project report template for details.)
Moving Toward a New Era of Precision Breast Cancer Treatment
Improved access to innovative drugs is reshaping the landscape of breast cancer diagnosis and treatment. Genetic testing, especially comprehensive and efficient NGS testing, is the core bridge connecting patients to precision treatment. It moves treatment away from a "one size fits all" approach and toward a truly "individualized" model.
In the future, as more innovative oncology drugs are developed and launched, the clinical value of genetic testing will become even more prominent. YunKang will continue to dedicate itself to technological innovation and service optimization, collaborating with clinical experts to promote the standardization and practical application of precision breast cancer diagnosis and treatment, lighting new hope for more patients.
Precise testing is the first step in pointing the way for treatment.
Choosing professionalism is the way to win more possibilities for life.
Copyright 2023 Yunkang Group Limited. All rights reserved. 粤ICP备12059545号-6 Privacy Policy | Disclaimers 网站建设技术支持:唯引互动

















