Recently, the 2025 results of multiple inter-laboratory quality assessments (EQA) for pathogen detection have been announced one after another. YunKang Group · Guangzhou DaAn Clinical Laboratory achieved outstanding performance, not only passing the National Center for Clinical Laboratories (NCCL) bacterial resistance gene detection专项 assessment with high scores, but also obtaining "all excellent" results in the assessments organized by the Shanghai Center for Clinical Laboratory (SCCL). These assessments covered core scenarios including respiratory tract infections, fungal detection, and tuberculosis resistance analysis, demonstrating the standardization and precision of YunKang’s tNGS technology with solid strength.

Shanghai Center for Clinical Laboratory Series EQA
2025 tNGS Detection of Lower Respiratory Tract Pathogens (Survey Project)
In the SCCL series EQA, YunKang Laboratory demonstrated excellent detection sensitivity and specialized testing capabilities. For the lower respiratory tract pathogen detection project, which required detecting various pathogens at levels as low as 10 copies/mL, YunKang Laboratory, relying on optimized testing workflows and data analysis algorithms, successfully achieved accurate detection of all samples, fully showcasing the robust capability of tNGS technology in detecting low-concentration pathogens.
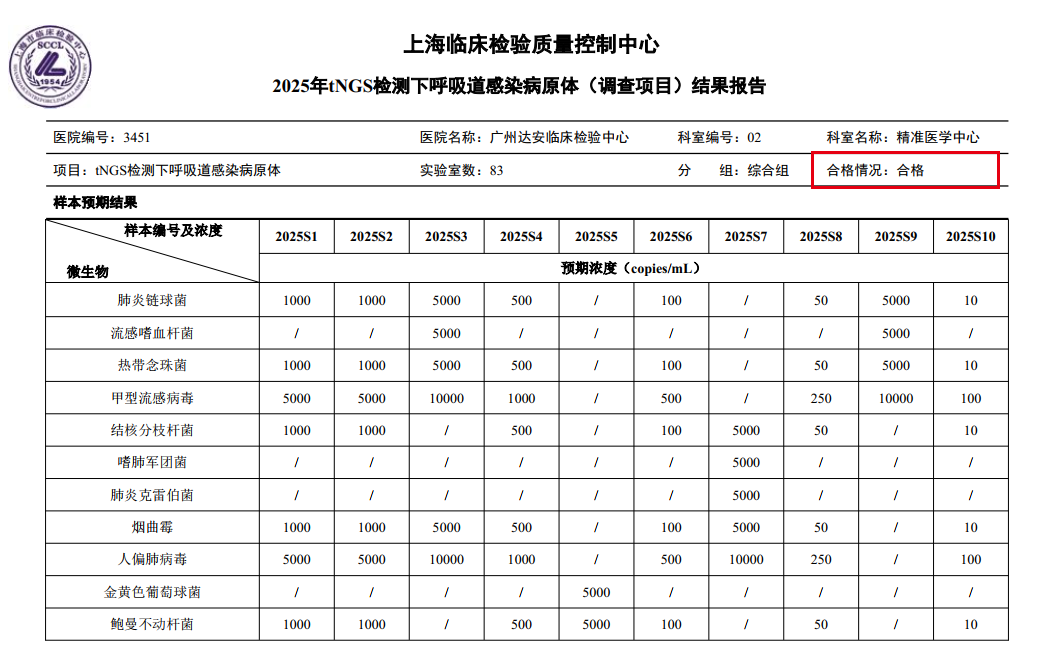
SCCL Lower Respiratory Tract tNGS Results Report
2025 Detection of Aspergillus spp., Cryptococcus neoformans, and Pneumocystis jirovecii Nucleic Acids (Survey Project)
Facing the industry challenge of detecting nucleic acids from Aspergillus spp., Cryptococcus neoformans, and Pneumocystis jirovecii – where nucleic acid extraction efficiency from filamentous fungi and cryptococci is low and the limit of detection is far lower than for common bacteria – YunKang participated in this project using tNGS technology and passed with a perfect score, highlighting YunKang’s tNGS capability in detecting key and difficult pathogens such as fungi.

SCCL Results Report for Aspergillus spp., Cryptococcus neoformans, and Pneumocystis jirovecii Nucleic Acid Project
2025 Mycobacterium tuberculosis Rifampicin Resistance Detection (Survey Project)
YunKang participated in this EQA using its Panoramic Targeted Next Generation Sequencing (tNGS) for Mycobacteria assay. The detection results for rifampicin resistance mutations were completely consistent with expectations.
This assay can detect 9 species and variants of the Mycobacterium tuberculosis complex, 183 species or subspecies of non tuberculous mycobacteria, 48 coinfection / differential diagnosis related pathogens, and covers 38 drug resistance genes (with over 3,500 mutation sites) for first and second line anti TB drugs as well as NTM related drugs, providing strong support for clinical diagnosis and resistance assessment of mycobacterial diseases.
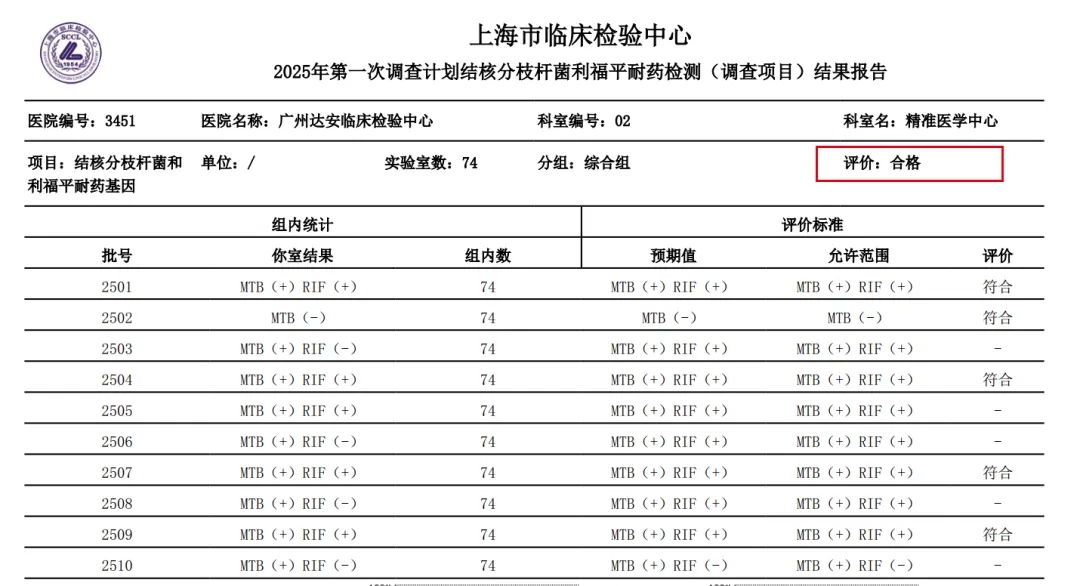
SCCL Results Report for Mycobacterium tuberculosis Rifampicin Resistance Detection
National Center for Clinical Laboratories EQA
In the national level EQA, YunKang also performed brilliantly. It achieved perfect scores in multiple NCCL bacterial resistance gene detection projects, including genotyping of oxacillin resistant Staphylococcus aureus, genotyping of vancomycin resistant Enterococcus, enzyme type genotyping of carbapenem resistant Enterobacteriaceae, and detection of Mycoplasma pneumoniae nucleic acid and its resistance genes.

These assessments covered common clinically relevant resistant bacteria and key resistance gene loci. YunKang’s perfect score performance confirmed the high sensitivity and accuracy of its tNGS technology in resistance gene detection, enabling rapid molecular drug susceptibility assessment results for clinical use and supporting precise adjustment of medication regimens.
In addition, YunKang’s tNGS technology successfully passed two additional EQAs for HPV genotyping and HPV E6/E7 gene expression, continuously expanding the application boundaries of the technology.
This "Grand Slam" certification fully affirms YunKang’s capabilities in tNGS technology R&D, standardized process construction, and quality control systems.
In the future, YunKang will continue to deepen its efforts in the field of pathogenic microorganism detection, providing more accurate and efficient testing solutions to support rapid diagnosis and precision treatment of infectious diseases through technological innovation and service upgrades.
Copyright 2023 Yunkang Group Limited. All rights reserved. 粤ICP备12059545号-6 Privacy Policy | Disclaimers 网站建设技术支持:唯引互动
















